
Converting Between Particles and Moles Using Avogadro's Number and Conversion Factors – Science and Joe
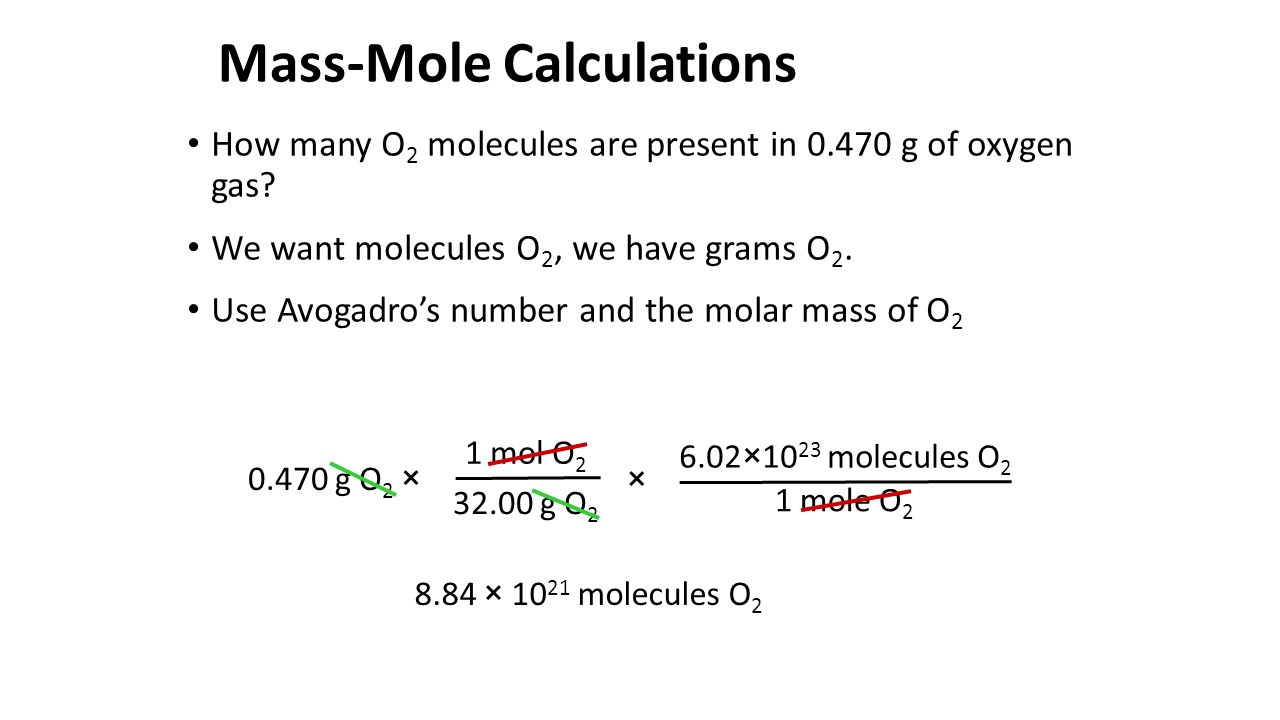
The Mole Concept. Avogadro's Number Avogadro's Number (symbol N) is the number of atoms in grams of carbon. Its numerical value is 6.02 × ppt download

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
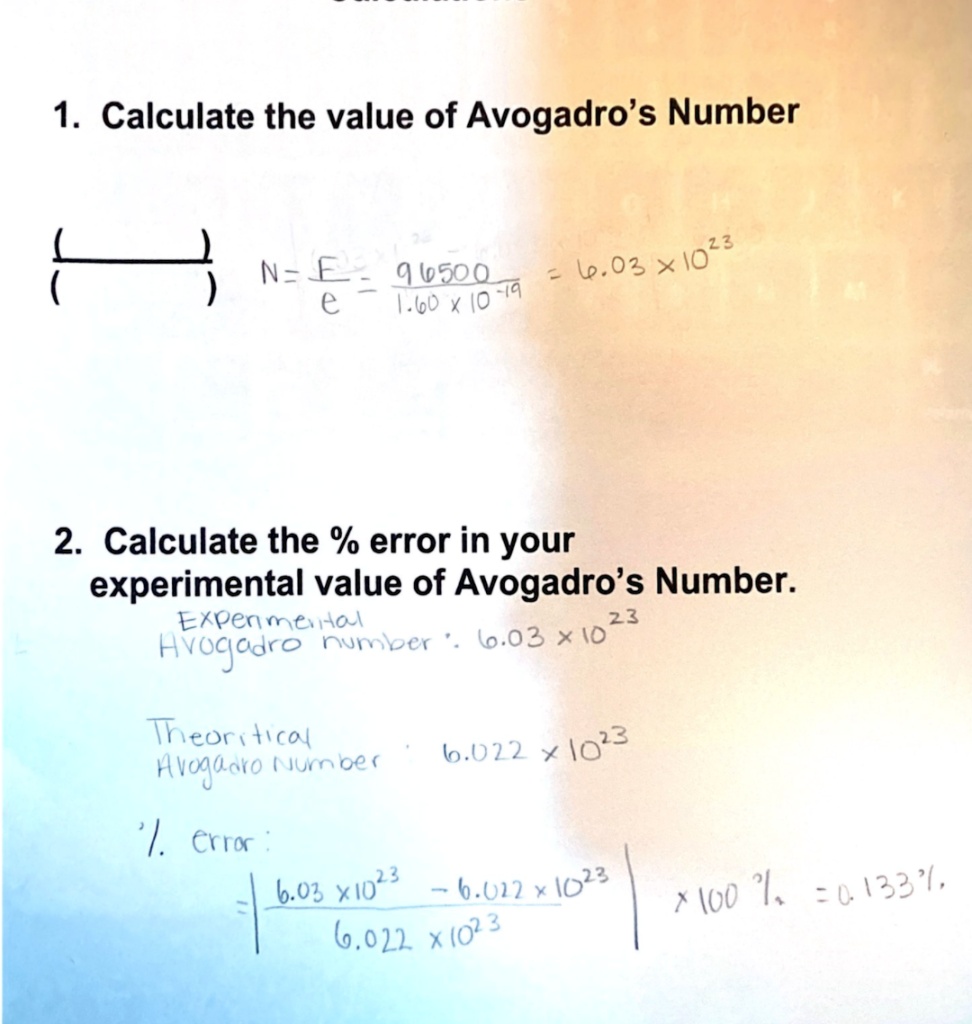
SOLVED: 1 Calculate the value of Avogadro's Number 13 l.03 x10 1 N= € 9.k5oo A.6o / I0 Fiq 2. Calculate the % error in your experimental value of Avogadro's Number: Ekpen

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
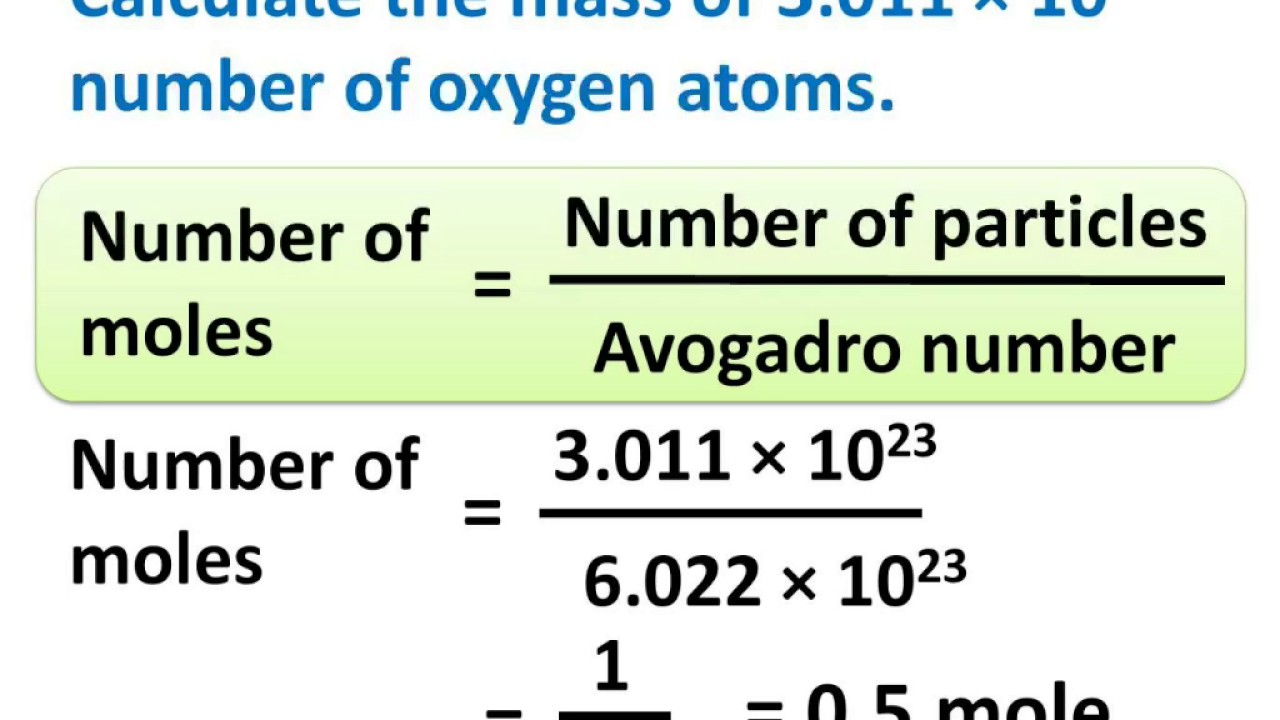

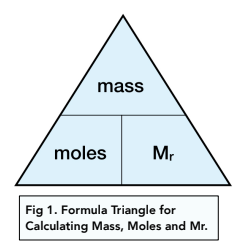



:max_bytes(150000):strip_icc()/Avogadro-58f7d6f35f9b581d5983024e.jpg)




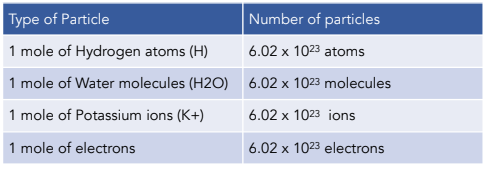









.PNG)